[edit]
Report on Face Masks for the General Public - An Update
Prepared for the DELVE Initiative by
Citation
(2020), Report on Face Masks for the General Public - An Update. DELVE Addendum MAS-TD1. Published 07 July 2020. Available from https://rs-delve.github.io/addenda/2020/07/07/masks-update.html.
Summary
The DELVE masks report1 summarised the available evidence suggesting that universal use of masks by the public in situations where physical distancing was not possible or predictable would decrease onward transmission of SARS-CoV2 from asymptomatic and presymptomatic infected individuals. Since this report was published, the UK government has mandated mask use on public transport and in health care settings. The British Medical Association has recommended the use of masks in these settings.2 The WHO has also revised its guidelines along these lines.3 Multiple countries, states and cities throughout the world have mandated mask use in all settings where distancing is not possible or predictable.4 The UK government has recommended but not mandated mask use in these settings. This update presents important new evidence that further supports the universal use of masks, including cloth masks in all settings where distancing is not possible or predictable. Universal mask use, including cloth masks, as a low cost way to facilitate the opening up of society is strongly supported for two reasons. First, prolonged contagiousness (median time 4 days)5 of asymptomatic infected people means that there is a high potential for disease transmission at a time when they are actively engaging with others. Second, there is increasing empiric evidence that mask use prevents SARS-CoV-2 transmission. The cumulative evidence, plus the recent change in physical distancing policy, strongly supports the use of masks in all circumstances where physical distancing of more than 1 meter cannot be maintained, and no physical barriers exist, including in shops, office buildings and public settings.
The original report was published on May 4, 2020.1 This paper has drawn on evidence available up to 28 June 2020 and has not been subject to formal peer-review. Further evidence on this topic is constantly published and DELVE may return to this topic in the future. This independent overview of the science has been provided in good faith by subject experts. DELVE and the Royal Society accept no legal liability for decisions made based on this evidence.
Table of Contents
Key points
-
Longitudinal data support initial findings that the majority of SARS-CoV2 infected individuals remain asymptomatic throughout infection. Even individuals who become symptomatic can have a several-day asymptomatic incubation period. Multiple new studies suggest that transmission of SARS-CoV2 by asymptomatic individuals is a substantial driver of transmission, including through droplets, aerosols and super spreader events.
-
Evidence continues to accrue that masks, including cloth masks, prevent onward transmission of infection. This is based on observational and modeling data in humans, on the effectiveness of masks in intercepting droplets and aerosols and on controlled studies of experimental animals. Evidence is also accumulating that masks may additionally protect the wearer.
-
There are no human controlled trials on the efficacy of universal mask use in stemming SARS-CoV2 transmission. It is inherently problematic to conduct such a trial on a practice that all must adhere to for it to be efficacious. Nevertheless, the cumulative multifaceted evidence on mask efficacy provides strong support for their use. Several accepted medical and public health practices to reduce the transmission of other infections are based on similar types of evidence and not on the results of randomized clinical trials.
-
New evidence finds that the use of clear plastic face shields can prevent onward transmission of droplets and aerosols. This addresses concerns from individuals and communities who/that cannot use masks, e.g., deaf people and individuals with breathing disorders,
-
While the UK government has mandated wearing of masks on public transport and in healthcare settings, it has not mandated mask use in other settings where the risk of transmission is high, such as in shops and close-quarter workplaces. This is concerning at a time when lockdown is being ended and the 2m distancing rule is being relaxed. The accumulated evidence warrants urgent reconsideration of government policy on masks. Mandatory use of masks or shields in all situations outside the home where physical distancing is not possible will help to counterbalance the expected increase in population-level exposure as lockdown ends.
Evidence Base
Evidence of Asymptomatic Infection and Transmission
Transmission of Covid-19 from asymptomatic or presymptomatic people allows community and institutional spread. These people, who are contagious, feel well and therefore may expose others. Multiple studies have shown that respiratory tract viral loads are highest for a few days preceding symptom onset and a few days afterwards. New evidence from epidemiological and modelling studies continues to lend strong support for the role of asymptomatic and pre-symptomatic transmission. Concurrently, there are new rigorous longitudinal data showing that the majority of SARS-CoV-2 infected individuals (up to ~ 90%) remain asymptomatic throughout infection as well as multiple new studies that add further support to asymptomatic transmission of SARS-CoV-2 being a substantial driver of transmission, including through aerosols and superspreader events.5 6 7 8 9 10 11 12 13
A recent longitudinal study highlights the finding that most infected individuals remain asymptomatic throughout the course of infection while carrying the virus for several days in their respiratory mucosa without ever manifesting symptoms.5 Investigation of the Covid-19 outbreak on the Diamond Princess cruise ship by Japanese public health officials resulted in the hospitalization of 96 asymptomatic infected people and 32 of their PCR-negative cabinmates, eight of whom later tested positive (though two were lost to follow-up). The individuals were monitored closely for symptoms, and they were re-tested by PCR between 5-7 days after the first test, and then on every 2-3 days. From the 104 asymptomatic infected individuals who were followed, 11 (10.6%) developed symptoms with a median asymptomatic incubation period of 4 days (interquartile range 3-5, range 3-7). The remaining 91 (89.4%) cleared the virus as defined by two consecutive negative PCR tests while remaining asymptomatic. 52% of them were still PCR-positive at 8 days and 10% at 15 days. Thus, asymptomatic infected individuals can have nasopharyngeal viral carriage for several days.
The devastating effect of presymptomatic transmission is highlighted by a report of a Covid-19 outbreak at a Seattle care home.6 A high rate of presymptomatic disease in care home residents allowed spread of infection to several facility wings. A total of 57 residents (64% of all residents) and 26 staff (18% of all staff) were infected. Infected staff included nurses, as well as physical therapists, food delivery staff and janitorial staff, many of whom worked in multiple facility wings. Eleven of the infected residents were hospitalised and 15 died. The lack of recognition of presymptomatic spread of infection, and the assumption that asymptomatic persons were not contagious, was the main factor that was linked to spread of infection in the facility.
New studies providing support for efficacy of masks in preventing onward transmission of SARS-CoV2
Observational Studies
-
A retrospective cohort study of households carried out over a 4 week period from 28 February to 27 March, 2020 in Beijing, China supports the efficacy of face masks (unstated type) in prevention of SARS-CoV2 transmission from asymptomatic/presymptomatic individuals in indoor settings.14 The secondary attack rate in families was 23.0% (77/335). Face mask use by the primary case and family contacts before the primary case developed symptoms was 79% effective in reducing transmission in comparison to controls. In a multivariable regression logistic model four factors were significantly associated with household transmission: diarrhea in the index case (OR 4.10, 95% CI 1.08 to 15.6); close contact within 1 meter to the index case for 1-3 close contacts (3.3, 1.05 to 10.40) and for ≥ 4 contacts (18.26, 3.93 to 84.79); ≥ 1 family member wearing a mask at home before disease onset in index case (0.21, 0.06 to 0.79); and surface disinfection ≥ 1 times/day (0.23, 0.07 to 0.84).
-
A recent study from Hong Kong supports the role of masks in preventing indoor transmission. All 14 of the case clusters occurring over the first 100 days of the pandemic occurred in indoor settings.15 Eleven of these clusters, that involved 113 cases, occurred in recreational mask-off settings (bars, restaurants, gyms) whereas there were only 3 clusters involving only 11 cases in workplace “mask on” settings (p = 0.04). The authors also surveyed mask-wearing compliance of 10,050 pedestrians in early April and found that 97% of them were wearing masks. They make the argument that one of the reasons for almost complete control of the epidemic in Hong Kong is because of universal mask wearing. The city is very densely populated, yet despite this has had an early and sustained control over transmission with only 148 cases total per million population and < 25 cases/day for the last several weeks.16
-
Mitze and colleagues used the early adoption of a universal public mask requirement in Jena, Germany to estimate mask effectiveness in abrogating transmission of SARS-CoV-2.17 They compared the Covid-19 incidence in Jena to other German cities and regions, controlling for other mitigation and host risk factors, using a “synthetic control” method in a carefully performed study of a natural experiment. They estimated that the mask wearing policy resulted in a 40 to 60% decrease in the daily growth rate of the epidemic. The estimated effect of a universal mask wearing policy was larger for larger cities than for the entire region studied. A limitation of the study is the lack of uncertainty for their estimates. Of note, this study shows the effect of a compulsory mask policy and not mask wearing per se, since no data are presented on compliance with the policy or the type of mask worn. The German mask policy did not specify the type of mask to be used. It stipulated that the mask/face covering used had to cover the mouth and nose.
Modeling Studies
-
Stutt and colleagues model the effectiveness of masks in preventing transmission of Covid-19, with assumptions of variable mask effectiveness, mask wearing compliance and varying importance of non-airborne infection.18 They conclude that disease transmission can be markedly decreased over a plausible range of mask effectiveness with widespread use of masks by the public. A limitation of this study is that it is strictly theoretical and therefore the conclusions depend on the base assumptions of mask effectiveness and constant mask use at all times when proximate to another person for optimal effectiveness.
-
Ngonghala and colleagues19 and Eikenberry and colleagues,20 modeled the impact of mask wearing on epidemic growth of Covid-19, as well as hospitalizations and mortality. These two studies are mainly from one academic department and use assumed values of mask effectiveness and compliance. The data are useful to visualize the relative impacts of mask effectiveness and mask wearing compliance on disease transmission and outcomes. They provide support for widespread mask wearing even if the estimated effect sizes may not be accurate. Apart from the limitation of this being a purely modeling study with assumed parameters that may or may not be correct, it does not factor in the possibility of disease transmission by direct inoculation of mucosal surfaces with virus-containing respiratory secretions.
-
Zhang and colleagues estimated that public wearing of masks resulted in tens of thousands fewer cases of Covid-19 infection in New York City and elsewhere.21 They also specify that disease transmission is mostly via aerosols. However, the conclusions of this study are not supported by the presented evidence, as summarized by a number of scientists requesting a retraction by the authors.22
New observation study providing support for efficacy of masks in protecting the wearer from SARS-CoV2
- A Covid-19 outbreak occurred on the United States Naval aircraft carrier, the USS Roosevelt.23 Approximately 25% of its 4200-member crew were affected over the course of a three-month voyage from January to April 2020. Respiratory illnesses can spread easily on naval ships because of tight living quarters and close contact between crew members.24 Of 382 crew members surveyed, 60% had evidence for Covid-19 infection. A face covering was worn by 81% of those not developing infection, and 56% of those who became infected. Thus, use of a face covering was significantly associated with a decreased risk of becoming infected (OR 0.3, 95% CI 0.2 to 0.5). This study suggests that face coverings can also provide protection to the wearer.
Surgical and cloth masks reduce onward transmission of small particle aerosols of infectious agents
Several clinical and laboratory studies demonstrate that surgical and cloth masks can effectively reduce the transmission of small particle aerosols of infectious agents. Since aerosol particles are smaller than airborne droplets, this substantiates the effectiveness of surgical and cloth masks for source control of droplets.
-
Mycobacterium tuberculosis, the bacterium causing tuberculosis, is a prime example of an infection spread predominantly by the aerosol route from the cough of TB patients. Dharmadhikari et al. showed that wearing a surgical mask by patients with pulmonary TB reduced aerosol transmission to guinea pigs placed in the room exhaust air. 69 of 90 (77%) guinea pigs exposed to air from unmasked patients developed TB versus 36 of 90 (40%) animals exposed to air from masked patients, a 52% reduction (95% CI 40 - 63%).25 Cough is a prominent feature of lung TB and guinea pigs are exquisitely sensitive to TB. Thus, surgical masks were efficacious in preventing airborne transmission not only from breathing and talking, but also from coughing. Indeed, TB patients are routinely asked to wear surgical masks (rather than N95 masks) to prevent onward transmission.26
-
Pseudomonas aeruginosa is a bacterium that colonizes and causes pathology in the lungs of cystic fibrosis patients and can be spread by aerosols. Two studies show the efficacy of surgical masks in preventing transmission of P. aeruginosa aerosols.27 28 In the first study, wearing surgical masks and covering the mouth when coughing reduced the production of P. aeruginosa aerosols by 90% in patients with cystic fibrosis during coughing and by 95% during talking when sampled at a 2m distance.27 In this study, 71 to 86% of captured aerosol P. aeruginosa particles from subjects not wearing masks while coughing were <4.7 um in diameter. The second study also showed that surgical masks reduced aerosol (<4.7 um) transmission of P. aeruginosa from coughing CF patients by 86% in comparison to no masks.28
-
A series of controlled experiments in a hamster SARS-CoV2 transmission model provides a strong case for the use of surgical masks in reducing transmission, particularly for source protection from the infected case.29 Infected (“index”) hamsters were co-housed separated by barriers such that they shared the same closed airspace, but no physical contact was possible. In two of the three groups the barriers were covered with standard surgical mask material such that the fluid repellant exterior mask surface was facing either the uninfected animals (simulating protection from onward transmission) or the index hamster (simulating masking the infected subject). Non-contact transmission was observed in 10 of 15 (67%) of the unprotected animals, 2 of 12 (16.7%) animals when the index animal was “masked”, and 4 of 12 animals where the naïve animals were “masked”. The odds ratios of infection compared to unmasked controls were 0.1 (95% CI 0.02 to 0.64) and 0.25 (0.05 to 1.2) for “masking” of the infected animal and “masking” of the uninfected animals, respectively. This represents a 75% reduction in transmission when the infected animal was “masked”, and a 50% reduction when the uninfected animals were “masked”. Neither of the masked groups mounted a Covid-19 antibody response whereas all of the index hamsters and 60% of the unmasked exposed hamsters did, and regardless of mask orientation all mask protected animals were significantly less ill than non-mask-protected animals.
Thus, surgical masks can prevent transmission of two bacterial lung infections known to be transmitted by aerosol, by intercepting the aerosolized bacteria even from coughing patients. An animal model study suggests the same is true for SARS-Cov2. Therefore, for those SARS-CoV2 transmission events that are aerosol-transmitted masks could provide excellent source control.
Cloth masks can prevent transmission of aerosols and droplets
The worldwide push to use nonmedical cloth masks led us to review a new study on this topic, and importantly to revisit studies on the efficacy of surgical masks from a time when they were made of cloth and reused.
-
An in-silico study that modeled the utility of cloth facemasks concluded that cloth masks should provide excellent source protection from coughing or sneezing, where they would greatly reduce the turbulent flow that can otherwise send small droplets for distances of 7-8 meters.30 They predicted two different effects depending on droplet size, completely stopping (absorbing) those 4 um or greater, while reducing the velocity of the air carrying the smaller ones. Their modeling results are in general agreement with experimental studies cited in the main report.
-
Two older studies suggest the efficacy of cloth “surgical” masks for source protection.31 32 Until at least the mid-1970’s surgical masks were made of cloth, usually cotton, layers. Both show that these reusable cloth masks were 90 to 95% efficient at filtering outward oral-origin aerobic bacterial particles in the 1-10 um diameter range.
-
An important paper from 1978 examined the efficacies of different weaves of cotton as well as the incorporation of a non-woven fabric layer in protection against aerosols.33 Using an aerosol of a bacterial suspension in saline, cotton materials had filtration efficacies of 43-94% depending on the weave. The filtration efficacies were 43% for bleached cotton (thread 40x46), 69% for bleached cotton with an increased thread (46 X50), 73% for Calico (thread 80 X 80) and 94% for Twill weave cotton. Fine glass fiber with nonwoven fabric had 98% - 99% effectiveness. A noteworthy aspect of this paper is that in 1978, there already existed a standard commercial machine for testing materials with aerosols.
-
A study of tightly fitting cloth masks or respirators examined the protection of experimental animals from aerosolized Mycobacterium tuberculosis. Cloth masks (6 layers, 44 thread/inch) performed almost as well as the respirators, preventing 77% of infections versus 97% for the respirators.34 While this was not a study of source control, but rather of wearer protection, it demonstrates that cloth masks can prevent the entry of particles that are smaller than the holes between the mask fibers, likely because of overlapping layers that produce an irregular path for particles. It demonstrates that mask protection is dependent not only on the fabric type but also on its fit, and that multiple cloth layers can be very effective at filtering small (< 5 um diameter) particles.
Characteristics of cloth masks
As mask use is becoming widespread, it is important to identify materials that will prevent droplet transmission and yet be comfortable to wear. Cloth masks can be made that have high filtration efficiency, but as this increases the resistance to airflow through the mask also increases. A metric taking both factors into account is termed the filter quality factor (Q factor), and is used as a single measure of wearability and protection, based on U.S. National Institute for Occupational Safety and Health standard tests for masks.35 The challenge for cloth mask manufacture is to optimize the Q factor, and the fit of the mask. Several modifications of cloth masks can increase filtration without excessively increasing flow resistance. These modifications include the use of an electrostatically charged fabric,35 layering of materials and use of different materials in the mask36 37 and increasing fabric hydrophobicity.38
Since all of these in vitro studies assume 100% fit, which is not the case for all but properly fitted respirators such as the FFP2 or N95 masks, the actual filtration effectiveness is only a rough guide to the use of these fabrics in mask construction.39 Facial fit, including the use of nose clip, is likely as or more important than the fabric used in the mask. Of vital importance is that prevention of transmission from an infected source will not be reliable if exhaust valves are built into a mask. Because cloth mask demand will increase with a requirement for their wear on public transport and for hospital visitors, consideration should be given to establishing basic manufacturing standards and testing guidelines. The Association Française de Normalisation (AFNOR) has established standards and guidelines for such masks, which they term barrier masks.40 41 The AFNOR standards prohibit both intake and exhaust valves. Of note, the AFNOR standards concern primarily wearer protection as opposed to prevention of source transmission, so any new UK standards will have to consider the prevention of source transmission as well. Standards like this will help the UK fabric industry meet demand to make optimally performing cloth masks. At least one UK manufacturer has already begun this process.42
Studies of air flow patterns around masks
-
Dbouk and Drikakis report the theoretical distribution of respiratory droplets produced by coughing and their interruption by wearing a theoretical mask.43 44 These papers provide a theoretical framework for mask effectiveness and droplet spread, show that leaks around the mask need to be considered, and that a mask reduces the velocity of forward spread.
-
Tang and colleagues demonstrated qualitatively the ability of a surgical mask to interrupt turbulent air resulting from a cough as well as leakage around the mask, providing supporting evidence for the Dbouk and Drikakis studies.45
-
Viola and colleagues provide qualitative and quantitative data on the ability of N95, surgical and cloth face masks, and face shields, to block forward transmission during coughing and breathing, amplifying on the Tang study.46 They found that 90% of forward transmission was blocked by all mask types, including the homemade cloth masks. Depending on the gap between the covering and the face there was downward, lateral, upward and backward leakage.
Face shields for individuals unable to wear masks
Face masks present problems for certain individuals, e.g. people with chronic respiratory conditions like asthma and deaf people who rely on lip reading.16 47 Clear plastic face shields could circumvent the difficulties with mask use in these populations. Studies on face shields are more limited and address protection to the recipient rather than their ability to prevent onward transmission.48 One recent study measured air (with an optical technique), coming from breathing and coughs found shields highly effective (more than masks) at stopping direct frontward emission, although most of the air was sent downward, and smaller amounts to the sides and upward.39 Therefore, shields would be unsuitable to prevent transmission to people lower than the wearer, e.g. sitting on a bus next to a standing wearer or lying down in care-settings. For two standing people of similar height facing one another, protection would be very good, but aerosols would still get dispersed in an enclosed space. However, leakage jets were also seen with masks, including downward and to the back and sides.
Definition of Terms
Airborne transmission Transmission of an infectious agent through the air, from an infected source to an uninfected person. This includes both droplet and aerosol transmission.
Aerosol route of infection Airborne transmission of small particles (generally ≤ 5 um diameter) that can enter the lung alveoli directly during inhalation, and can persist suspended in the air for hours. Examples of infections that are transmitted by aerosols include measles and tuberculosis.
Droplet route of infection Airborne transmission of large particles (generally ≥ 10 um diameter) that because of their size cannot easily enter the lung alveoli. These particles fall to the ground within minutes, traveling short distances (usually 1 to 2 meters) before falling. They cause infection by deposition in the nose, mouth or eye. The primary infection is therefore of the eye (conjunctivitis), the upper respiratory tract (rhinitis, pharyngitis) or both sites. Those infectious agents that spread by the droplet route and cause pneumonia do so in a sequential fashion with the initial infection being in the upper respiratory tract and a secondary infection in the lung. Coronavirus infections are transmitted primarily by the droplet route.
Super spreader event Transmission of airborne infection from a single person to a large number of people at one time and place.
Mask A protective covering of the mouth and nose. Surgical and medical masks are made of synthetic fabrics, and are usually fluid repellant at the outer face. Masks are distinguished from respirators in that the former are relatively loosely fitting and the latter form a tight seal around the face or head. Examples of respirators are the N95, FFP2 and powered air purifying respirator (PAPR). Cloth masks are a type of mask and can be used for similar purposes, depending on design.
Cloth face covering A type of mask made from cloth materials of one to several layers, varying in design and fit. An alternative term used to distinguish these masks from certified medical or surgical masks.
Onward transmission Transmission of an infectious agent from an infected person to an uninfected person. (Onward transmission and Source control are converse terms.)
Source control Prevention or reduction of transmission of infectious agent from an infected person to an uninfected person. (Source control and Onward transmission are converse terms). See Figure.
Wearer protection Prevention or reduction of deposition of infectious droplets into the mouth or nose of, or inhalation of infectious aerosols by, the wearer of a mask or cloth face covering. See Figure 1.
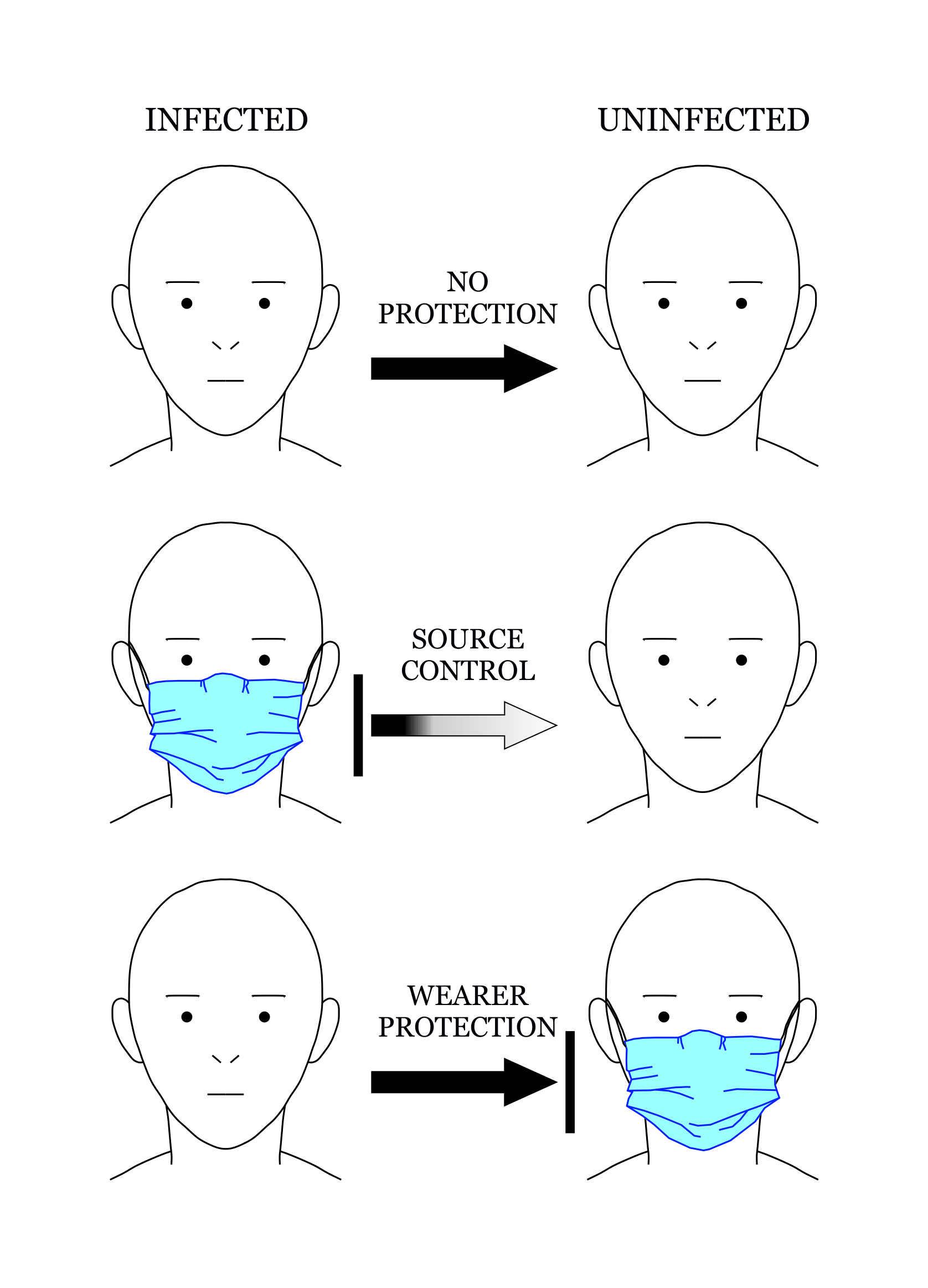
Figure 1. The use of masks and face coverings for source control contrasted with the use of masks for wearer protection.
Footnotes and References
-
DELVE (2020) Face masks for the general public. Available at: https://rs-delve.github.io/reports/2020/05/04/face-masks-for-the-general-public.html ↩ ↩2
-
BMA (2020) Face covering measures are important now and should not be restricted to public transport. Available from: https://www.bma.org.uk/news-and-opinion/face-covering-measures-are-important-now-and-should-not-be-restricted-to-public-transport-says-bma ↩
-
World Health Organization (2020) Advice on the use of masks in the context of COVID-19: interim guidance, 6 April 2020. Report No.: WHO/2019-nCov/IPC_Masks/2020.4. Available at: https://apps.who.int/iris/handle/10665/331693 ↩
-
Masks for all (2020) Available from: https://masks4all.co/ ↩
-
Sakurai, A., Sasaki, T., Kato, S., Hayashi, M., Tsuzuki, S.I., Ishihara, T., et al. (2020) Natural History of Asymptomatic SARS-CoV-2 Infection. N Engl J Med. doi: 10.1056/NEJMc2013020 ↩ ↩2 ↩3
-
Arons, M., Hatfield, K., Reddy, S., Kimball, A., James, A., Jacobs, J. et al. (2020) Presymptomatic SARS-CoV-2 Infections and Transmission in a Skilled Nursing Facility. N Engl J Med. doi: 10.1056/NEJMoa2008457; ↩ ↩2
-
Treibel, T., Manisty, C., Burton, M., McKnight, A., Lambourne, J., Augusto, J. et al. (2020) COVID-19: PCR screening of asymptomatic health-care workers at London hospital. Lancet. doi: 10.1016/s0140-6736(20)31100-4; ↩
-
Streeck, H., Schulte, B., Kuemmerer, B., Richter, E., Hoeller, T., Fuhrmann, C. et al. (2020) Infection fatality rate of SARS-CoV-2 infection in a German community with a super-spreading event. medRxiv. doi: 10.1101/2020.05.04.20090076; ↩
-
James, A., Eagle, L., Phillips, C., Hedges, D., Bodenhamer, C., Brown, R. et al. (2020) High COVID-19 Attack Rate Among Attendees at Events at a Church - Arkansas, March 2020. CDC Morbidity and Mortality Weekly Report. doi: 10.15585/mmwr.mm6920e2; ↩
-
Emery, J., Russell, T., Liu, Y., Hellewell, J., Pearson, C., CMMid Covid-Working G et al. (2020) The contribution of asymptomatic SARS-CoV-2 infections to transmission - a model-based analysis of the Diamond Princess outbreak. medRxiv, doi: 10.1101/2020.05.07.20093849v1; ↩
-
Prather, K., Wang, C., Schooley, R. (2020) Reducing transmission of SARS-CoV-2. Science. doi: 10.1126/science.abc6197; ↩
-
Adam, D., Wu, P., Wong, J., Lau, E., Tsang, T., Cauchemez, S. et al. (2020) Clustering and superspreading potential of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infections in Hong Kong. Research Square, doi: 10.21203/rs.3.rs-29548/v1; ↩
-
Miller, D., Martin, M., Harel, N., Kustin, T., Tirosh, O., Meir, M. et al. (2020) Full genome viral sequences inform patterns of SARS-CoV-2 spread into and within Israel. medRxiv. doi: 10.1101/2020.05.21.20104521 ↩
-
Wang, Y., Tian, H., Zhang, L., Zhang, M., Guo, D., Wu, W. et al. (2020) Reduction of secondary transmission of SARS-CoV-2 in households by face mask use, disinfection and social distancing: a cohort study in Beijing, China. BMJ Glob Health. doi: 10.1136/bmjgh-2020-002794 ↩
-
Cheng, VC-C., Wong, S-C., Chuang, VW-M., So, SY-C., Chen, JH-K., Sridhar, S. et al. (2020) The role of community-wide wearing of face mask for control of coronavirus disease 2019 (COVID-19) epidemic due to SARS-CoV-2. J Infect. doi: 10.1016/j.jinf.2020.04.024 ↩
-
Asthma UK (2020) What should people with asthma do now? How to cut the risk of getting coronavirus and what happens to your usual asthma care. Available from: https://www.asthma.org.uk/advice/triggers/coronavirus-covid-19/what-should-people-with-asthma-do-now/ ↩ ↩2
-
Mitze, T., Kosfeld, R., Rode, J., Wälde, K. (2020) Face Masks Considerably Reduce COVID-19 Cases in Germany: A Synthetic Control Method Approach. IZA – Institute of Labor Economics, Contract No.: IZA DP No. 13319. Available at: https://www.iza.org/publications/dp/13319/face-masks-considerably-reduce-covid-19-cases-in-germany-a-synthetic-control-method-approach ↩
-
Stutt, R., Retkute, R., Bradley, M., Gilligan, C., Colvin, J. (2020) A modelling framework to assess the likely effectiveness of facemasks in combination with ‘lock-down’ in managing the COVID-19 pandemic. Proceedings of the Royal Society A. doi: 10.1098/rspa.2020.0376 ↩
-
Ngonghala, C., Iboi, E., Eikenberry, S., Scotch, M., MacIntyre, C., Bonds, M. et al. (2020) Mathematical assessment of the impact of non-pharmaceutical interventions on curtailing the 2019 novel Coronavirus. doi: 10.1016/j.mbs.2020.108364 ↩
-
Eikenberry, S., Mancuso, M., Iboi, E., Phan, T., Eikenberry, K., Kuang, Y. et al. (2020) To mask or not to mask: Modeling the potential for face mask use by the general public to curtail the COVID-19 pandemic. Infect Dis Model. doi: 10.1016/j.idm.2020.04.001 ↩
-
Zhang, R., Li, Y., Zhang, A., Wang, Y., Molina, M. (2020) Identifying airborne transmission as the dominant route for the spread of COVID-19. Proceedings of the National Academy of Sciences. doi: 10.1073/pnas.2009637117 ↩
-
Streeck, H., Schulte, B., Kuemmerer, B., Richter, E., Hoeller, T., Fuhrmann, C. et al. (2020) Infection fatality rate of SARS-CoV-2 infection in a German community with a super-spreading event. medRxiv. doi: 10.1101/2020.05.04.20090076 ↩
-
Payne, D., Smith-Jeffcoat, S., Nowak, G., Chukwuma, U., Geibe, J., Hawkins, R. et al. (2020) SARS-CoV-2 Infections and Serologic Responses from a Sample of U.S. Navy Service Members - USS Theodore Roosevelt, April 2020. Morbidity and Mortality Weekly Report. doi: 10.15585/mmwr.mm6923e4 ↩
-
Sanchez, J., Cooper, M., Myers, C., Cummings, J., Vest, K., Russell, K. et al. (2015) Respiratory Infections in the U.S. Military: Recent Experience and Control. Clinical Microbiology Reviews, 28(3):743. doi: 10.1128/CMR.00039-14 ↩
-
Dharmadhikari, A., Mphahlele, M., Stoltz, A., Venter, K., Mathebula, R., Masotla, T. et al. (2012) Surgical face masks worn by patients with multidrug-resistant tuberculosis: impact on infectivity of air on a hospital ward. Am J Respir Crit Care Med, 185(10):1104-9. doi: 10.1164/rccm.201107-1190OC ↩
-
National Institute for Health and Care Excellence (2019) Tuberculosis: management and infection control in hospital. Available at: https://pathways.nice.org.uk/pathways/tuberculosis/tuberculosis-management-and-infection-control-in-hospital.pdf ↩
-
Wood, M., Stockwell, R., Johnson, G., Ramsay, K., Sherrard, L., Jabbour, N. et al. (2018) Face Masks and Cough Etiquette Reduce the Cough Aerosol Concentration of Pseudomonas aeruginosa in People with Cystic Fibrosis. American Journal of Respiratory and Critical Care Medicine, 197(3):348-55. doi: 10.1164/rccm.201707-1457OC ↩ ↩2
-
Driessche, K., Hens, N., Tilley, P., Quon, B., Chilvers, M., Groot, R. et al. (2015) Surgical Masks Reduce Airborne Spread of Pseudomonas aeruginosa in Colonized Patients with Cystic Fibrosis. American Journal of Respiratory and Critical Care Medicine. 192(7):897-9. doi: 10.1164/rccm.201503-0481LE ↩ ↩2
-
Chan, JF-W., Yuan, S., Zhang, A., Poon, VK-M., Chan, CC-S., Lee, AC-Y. et al. (2020) Surgical mask partition reduces the risk of non-contact transmission in a golden Syrian hamster model for Coronavirus Disease 2019 (COVID-19). Clin Infect Dis. doi: 10.1093/cid/ciaa644 ↩
-
Kumar, V., Nallamothu, S., Shrivastava, S., Jadeja, H., Nakod, P., Andrade, P., et al. (2020) On the utility of cloth facemasks for controlling ejecta during respiratory events. arXiv. Available from: https://arxiv.org/abs/2005.03444 ↩
-
Greene, V. and Vesley, D. (1962) Method for Evaluating Effectiveness of Surgical Masks. Journal of Bacteriology, 83(3):663-7. PMID: 13901536 ↩
-
Quesnel, L. (1975) The efficiency of surgical masks of varying design and composition. Br J Surg, 62(12):936-40. doi: 10.1002/bjs.1800621203 ↩
-
Furuhashi, M. (1978) A study on the microbial filtration efficiency of surgical face masks–with special reference to the non-woven fabric mask. Bull. Tokyo Med Dent Univ. 25(1):7-15. PMID: 343940 ↩
-
Abramson, S. (1956) Experimental air-borne infection; a comparison of different masks in the protection of rabbits against inhalation infection with tubercle bacilli. Am Rev Tuberc. 73(3):315-29, doi: 10.1164/artpd.1956.73.3.315 ↩
-
Zhao, M., Liao, L., Xiao, W., Yu, X., Wang, H., Wang, Q. et al. (2020) Household materials selection for homemade cloth face coverings and their filtration efficiency enhancement with triboelectric charging. Nano Lett. doi: 10.1021/acs.nanolett.0c02211 ↩ ↩2
-
Konda, A., Prakash, A., Moss, G., Schmoldt, M., Grant, G., Guha, S. (2020) Aerosol Filtration Efficiency of Common Fabrics Used in Respiratory Cloth Masks. ACS Nano. doi: 10.1021/acsnano.0c03252 ↩
-
Xiao, L., Sakagami, H. and Miwa, N. (2020) A New Method for Testing Filtration Efficiency of Mask Materials Under Sneeze-like Pressure. In Vivo. doi: 10.21873/invivo.11955 ↩
-
Li, Y., Wong, T., Chung, J., Guo, Y., Hu, J., Guan, Y. et al. (2020) In vivo protective performance of N95 respirator and surgical facemask. Am J Ind Med, 49(12):1056-65. Epub 2006/11/11. doi: 10.1002/ajim.20395 ↩
-
Viola, IM. (2020) Face Coverings, Aerosol Dispersion and Mitigation of Virus Transmission Risk. Available from: https://arxiv.org/abs/2005.10720 ↩ ↩2
-
Association Française de Normalisation (2020) AFNOR SPEC S76-001. Barrier masks: guide to minimum requirements, methods of testing, making and use, Association Française de Normalisation, p. 1-36. Available at: https://www.afnor.org/en/news/protective-masks-download-our-reference-document-for-free/ ↩
-
Association Française de Normalisation (2020) AFNOR Spec - Barrier masks V1.0 2020. Available at: https://masques-barrieres.afnor.org/home/telechargement?culture=en-GB&_ga=2.122524043.1856562393.1592355638-141405935.1592355638 ↩
-
English Fine Cottons (2020) Britspun Face Covers for the Nation 2020. Available from: https://www.englishfinecottons.co.uk/journal/uncategorized/britspun-face-covers-for-the-nation/ ↩
-
Dbouk, T. and Drikakis, D. (2020) On coughing and airborne droplet transmission to humans. Physics of Fluids, 32(5):053310. doi: 10.1063/5.0011960 ↩
-
Dbouk, T. and Drikakis, D. (2020) On respiratory droplets and face masks. Physics of Fluids, 32(6):063303. doi: 10.1063/5.0015044 ↩
-
Tang, J., Liebner, T., Craven, B., Settles, G. (2009) A schlieren optical study of the human cough with and without wearing masks for aerosol infection control. J R Soc Interface. Suppl 6:S727-36. doi: 10.1098/rsif.2009.0295.focus ↩
-
Viola, IM. (2020) Face Coverings, Aerosol Dispersion and Mitigation of Virus Transmission Risk. Available from: https://arxiv.org/abs/2005.10720 ↩
-
BBC News (2020) Coronavirus: Mask wearing ‘risks isolating’ deaf people. Available from: https://www.bbc.com/news/uk-wales-52659083 ↩
-
Roberge, R. (2016) Face shields for infection control: A review. J Occup Environ Hyg. 13(4):235-42. doi: 10.1080/15459624.2015.1095302 ↩